THIOTEQ™Metal
Why let valuable dissolved metals go to waste? Why mix toxic metal with a pile of gypsym? THIOTEQ™Metal technology recovers dissolved metals from liquids by the formation of high purity metal sulphides. These can either be marketed or else disposed off in a very compact form.
The required sulphide for this process is generated on-site in a bioreactor from a low cost sulphur source at ambient temperature and pressure.
Typical application range for economically attractive metal recovery
| Application | Range |
| Dissolved metal concentration | 50 to 5,000 mg/l |
| pH range | < 0 to 10 |
| Temperature range | 0 to 80ºC |
| Typical sulphide demand | 100 kg to > 20 tonne per day |
For the simultaneous removal of sulphate and the recovery of metals please refer to SULFATEQ™.
Compared to precipitation as metal hydroxides, THIOTEQ™ offers
- Lower metal effluent concentrations
- Revenue from metal sulphides: attractive ROI at metal concentrations > 0.1 g/l
- Capability to deal with complexes and most chelating agents
- Better settling and dewatering characteristics of the metal sulphide sludge
Sulphide precipitation would be applied widely if the high cost of transport, storage or production of sulphide on-site (as NaSH or H2S) did not hamper its practical application.
The THIOTEQ™Metal technology solves this problem with a biogenic sulphide generator that generates sulphide on-site and on demand under ambient conditions. All that is needed to enable production is elemental sulphur, a food source like alcohol and some fertilizer-like nutrients. The sulphide is transported from the generator to one or more contactors via a closed gas recycle.
Advantages of THIOTEQ™Metal compared to NaSH or ‘chemical H2S’
- Cost-effective sulphide source: lower cost per tonne of metal sulphide produced
- Safe process at ambient conditions
- On-site sulphide production: no transport and storage of hazardous sulphide
- Better selectivity of metals, higher product quality
- Flexible process: production naturally adjusts to demand
- Robust and proven process
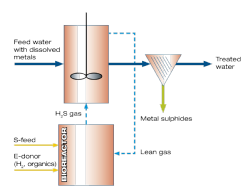
Working principle
The patented Paques THIOTEQ™Metal process consists of two stages: a chemical and a biological. The water to be processed only passes through the chemical stage. Sulphide is produced in the biological stage and transported to the chemical (precipitation) stage via a gas recycle.
Biological stage
A biocatalyst produces sulphide from elemental sulphur and an organic feedstock like ethanol. The biocatalyst grows in a bioreactor with a very long hydraulic retention time and is unaffected by the characteristics of the process stream. The conditions in the bioreactor are ambient, which makes it safe to operate.
Chemical stage - contactor
Sulphide is transported via a gas recycle to the chemical stage that consists of a gas-liquid contactor. The H2S dissolves in the process liquid and reacts virtually instantaneous with the dissolved metal. The metal sulphide that is formed is separated via gravity settling and dewatered. The lean gas is recycled back to the biological stage.
More information
For more information on Metal & Mining please contact us here and/or download our business case leaflet on how we recover copper at the Pueblo Viejo goldmine in the Dominican Republic.